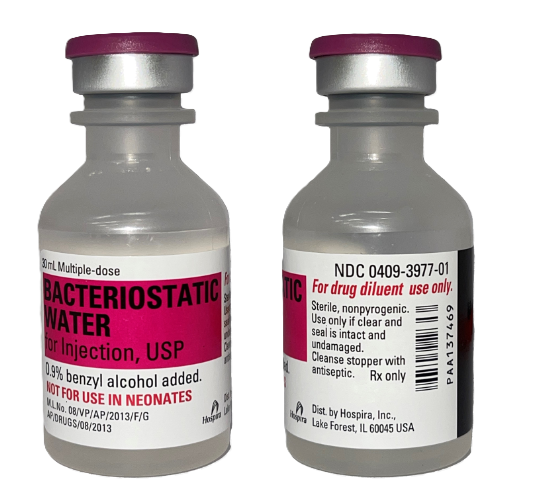
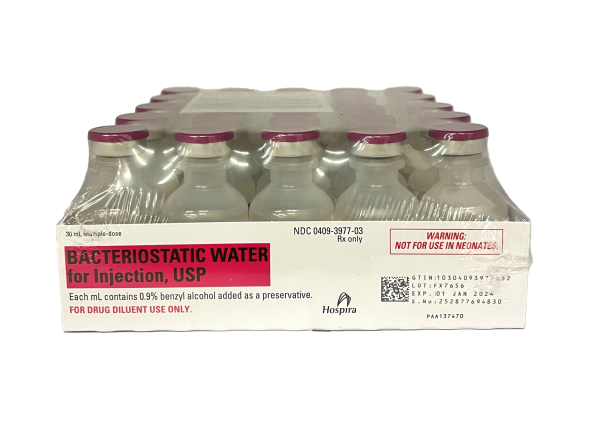
Bacteriostatic Water – Hospira Brand – 30ML Bottle
$30.00
Bacteriostatic Water (30mL) is a sterile, USP-grade solution containing 0.9% benzyl alcohol, designed for professional laboratory use in the reconstitution and dilution of research materials. Its bacteriostatic properties support safe multi-use access during controlled experimental procedures.
-
Ideal for reconstituting lyophilized peptides and research compounds
-
Contains 0.9% benzyl alcohol to support multi-use laboratory applications
-
Suitable for sterile dilution and solvent preparation in scientific testing
In stock

Volume Discounts
Bulk Volume Discounts starting as low as 20 units per Product

Custom Labeling
3 Tiers of labeling: Unlabeled, Generic, and Custom Label

Quality Guarantee
All products come with a 30-Day Quality Guarantee.

US-Based Support
US-based support available Monday - Friday 7-6pm ET
Description
Also Known As:
Bacteriostatic Water for Injection
CAS Number: 7732-18-5
Molecular Formula: H₂O
Molecular Weight: 18.02 g/mol
Form: Sterile Liquid Solution
Volume: 30mL vial
Composition: Water with 0.9% benzyl alcohol as a bacteriostatic preservative
Purity: USP-grade sterile solution
Product Overview
Bacteriostatic Water is a sterile, USP-grade diluent used in professional laboratory environments for reconstituting lyophilized peptides and other research materials. Formulated with 0.9% benzyl alcohol, it helps inhibit bacterial growth, allowing safe multi-use access from a single vial during ongoing experimental procedures.
This solution is widely utilized in scientific and analytical settings where controlled reconstitution, dilution, and solvent consistency are required.
Intended Applications
-
Reconstitution of lyophilized peptides for laboratory analysis
-
Dilution of research compounds in sterile environments
-
Multi-dose use requiring bacteriostatic preservation
-
General solvent for life-science and analytical testing
Storage & Handling
-
Store at controlled room temperature (20–25°C)
-
Use within 28 days after opening, following proper sterile technique
-
Do not freeze; protect from direct sunlight
⚠️ Professional Use Only – Important Notice
This product is intended strictly for Professional Use Only (PUO) in controlled laboratory, analytical, or scientific research settings. It is not for human consumption, medical administration, or therapeutic use of any kind.
All information provided is for scientific and educational purposes and has not been reviewed by the U.S. Food and Drug Administration. These materials are not intended to diagnose, treat, cure, or prevent any disease.
This product is not produced by or associated with any compounding pharmacy, outsourcing facility, or organization operating under sections 503A or 503B of the Federal Food, Drug, and Cosmetic Act.
Users are responsible for ensuring proper handling, storage, and compliance with all applicable regulations and professional standards.
Professional Use Only - Legal Disclaimer
All PeptiMed products are classified as Professional Use Only (PUO) and are intended exclusively for qualified professionals, licensed facilities, and authorized organizations. These products are not approved by the FDA and are not intended for personal use, consumer use, or unsupervised application.
By purchasing or using PeptiMed products, you confirm that you are a properly licensed professional acting within the scope of your credentials and that you understand all regulatory, safety, and compliance obligations associated with handling these materials. Products must not be sold, supplied, or provided directly to consumers or used outside an appropriate professional environment.
All handling, storage, and use of PeptiMed products are undertaken at your own risk. PeptiMed assumes no responsibility for misuse, improper application, or any clinical, regulatory, or professional consequences resulting from their use.
Labeling Options
All labeled vials include product details and batch information including a QR Code* to the COA printed on each label

Custom White-Label

Generic Label

Unlabeled Vial
*QR Codes coming in early 2026